With the 932 new positive cases of SARS-CoV-2 coronavirus detected this Sunday in Cuba, there are now 109,625 people diagnosed with this disease, (2.96%) since the outbreak of the pandemic in the country, on March 11, 2020, announced today the ministry of public health ( MINSAP by its Spanish acronym).
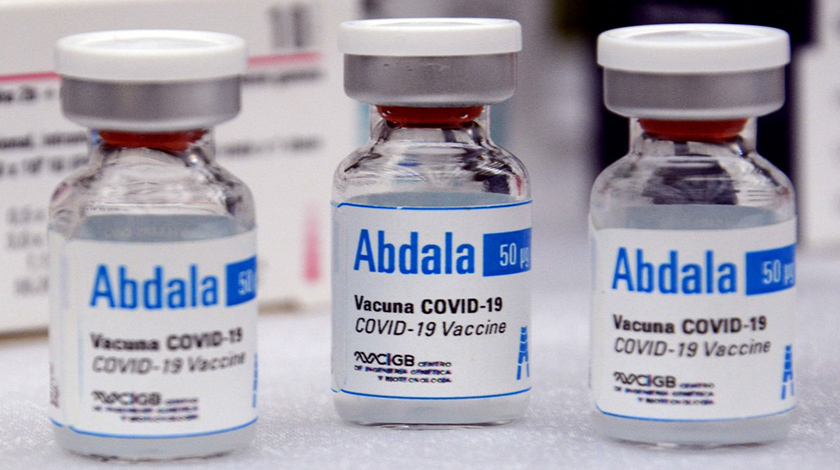
Cuban health authorities assess efficacy of vaccine candidate Abdala
Health professionals and the Center for Genetic Engineering and Biotechnology (CIGB) are assessing from today the efficacy of the Cuban anti-COVID-19 Abdala vaccine candidate, after concluding the application of the immunogen in the volunteers of the phase III clinical trial.
Cuba reports 932 new cases of COVID 19, 917 discharges and eleven deaths
Cuba studied 22,938 samples for COVID 19, resulting in 932 positive ones. The country accumulates 3, 700,029 samples taken and 109,625 positive.
CENCEC works to maintain certification of its quality management system
The National Coordinating Center for Clinical Trials (CENCEC) strives to keep complying with the Good Clinical Practices and to maintain its certification of its Quality Management System, in accordance with the requirements of the ISO 9001 Standard, a status it has held since 2008, granted by the National Bureau of Standards and AENOR International.
Genetic predisposition of ataxia patients to COVID-19 under study in eastern Cuba
A project to determine the genetic predisposition of patients with Spinocerebellar Ataxia type II (SCA2) to become infected with COVID-19 is under way by specialists in this province, top of the list in prevalence of this disease in Cuba.
Cuba reports 1,046 new cases of COVID 19, 973 discharges and twelve deaths
Cuba studied 22,451 samples for COVID 19, resulting in 1,046 positive ones. The country accumulates 3, 631,470 samples taken and 106,707 positive.
COVID-19 is affecting younger people and showing greater severity in hospitalized patients, expert says
Dr. Francisco Duran, national director of epidemiology of the Cuban ministry of public health ( MINSAP by its Spanish acronym) reported today in Havana that at the close of the last day there were 1,046 new cases of COVID-19 in Cuba and 12 deaths due to complications associated with the infection.
Community Population Studies expand in eastern Cuba due to COVID-19 spread
As part of the measures to tackle COVID-19 in the province of Holguin, the health system is expanding community population studies as part of the strategies to prevent infection with such a highly contagious virus.
Research on psychosocial risks of health personnel involved in the fight against COVID-19
A study by the National School of Public Health (ENSAP), together with the National Group of Health Psychology and other institutions, showed that our health workers are highly committed to, and happy to help in, the fight against COVID-19, a fact that favors work-related stress-response dampening.
Clinical trials of Cuban vaccine candidates proceed at a good pace
The administration of the final dose of Abdala in the eastern region and the third one of Soberana 02 in Havana, together with the development of intervention studies, mark the progress of clinical trials of the Cuban vaccine candidates toward their certification and the subsequent vaccination of the entire Cuban population.
Cuba produces 300 thousand doses of Abdala vaccine candidate every other day
Doctor Antonio Vallin Garcia, General Director of Aica Laboratories, said that between 280 and 300 thousand doses of the Abdala candidate are produced every other day and there are currently three batches on an industrial scale.
Cuban province readies for mass vaccination
Some 26 thousand inhabitants of the Cuban central province of Sancti Spíritus will be immunized against COVID-19 in the coming month of May, with the application of the Abdala vaccine candidate, developed by the Center for Genetic Engineering and Biotechnology of Havana.
Havana Opens New Health Facilities to Treat Increasing COVID-19 Cases
Havana authorities decided to increase the number of healthcare facilities to assist new COVID-19 cases and suspects of having the virus, in the face of a worsening spread of the coronavirus over the past weeks.
Cuban health authorities concerned about number of children infected with COVID-19
Experts from the Cuban ministry of public health (MINSAP by its Spanish acronym) warn about the high number of children, adolescents and young people who continue to be diagnosed with COVID-19, even in the midst of the restrictions and measures adopted in the country.
Hisplacen, 35 years leading the development of human placenta-derived products
Thirty-five years after its creation, the Doctor Carlos Miyares Cao Placental Histotherapy Center (Hisplacen) is consolidated as an international reference institution in the research, development, production and commercialization of medicines, cosmetics and nutrients derived from human placenta.