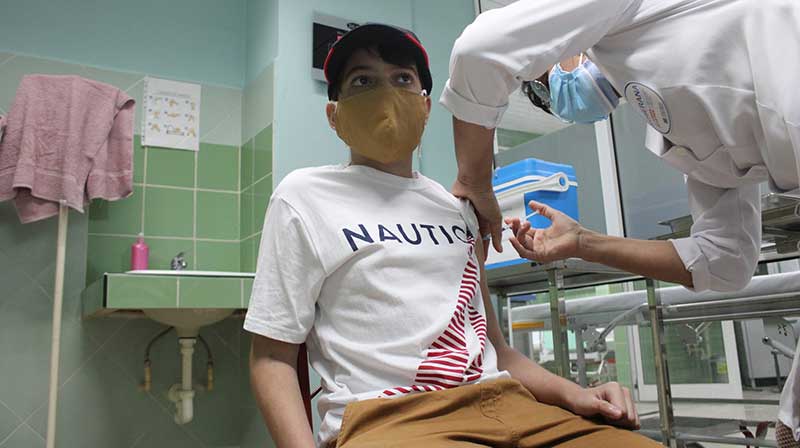
HAVANA, Cuba, Jun 18 (ACN) Since its approval on June 10 by the Center for State Control of Medicines, Equipment and Medical Devices (CECMED), the Soberana-Pediatrics clinical trial is progressing satisfactorily, said its leader, Dr. Meiby de la Caridad Rodríguez González, director of Clinical Research and Impact Evaluation of the Finlay Institute of Vaccines.
She pointed out that the first anti-COVID-19 research in children and adolescents complied with a rigorous approval process seven weeks after the dossier was submitted to the regulatory authority and on June 14, during the application of the first dose, they had the first sanitary inspection by the CECMED.
Soberana-Pediatrics includes in its phase I/II children and adolescents between three and 18 years of age, healthy, of Cuban nationality and 50 volunteers will be included in the first stage and 300 in the second, to whom the same scheme will be applied as for adults, that is, two doses of Soberana 02 and one of Soberana Plus, she pointed out.
The study will be carried out sequentially, after the first week of immunization, blood tests will be performed to determine the safety of the product, to request the approval of CECMED with a view to continue with the group from three to 11 years of age and to continue towards phase II with a greater number of adolescents.
The first stage of the research will be carried out at the Juan Manuel Márquez Pediatric Hospital and later the trial will be extended to the health areas of the municipalities of Playa, Marianao and La Lisa, she explained.
She said that it is a joy to evaluate the candidates in pediatric ages, because for 30 years the IFV has been producing preventive vaccines for this population group, and expressed her confidence in the quality of the candidates based on the scientific evidence demonstrated by its platform.
Sidebar

 Agencia Cubana de Noticias
Líder en información nacional
Agencia Cubana de Noticias
Líder en información nacional








Nos reservamos el derecho de no publicar los comentario que incumplan con las normas de este sitio