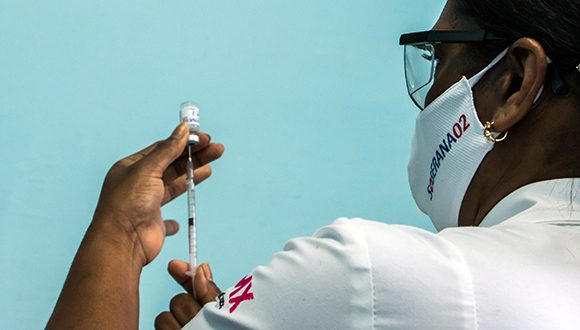
Havana, April 1 (ACN) The first round of phase III trials with the Cuban COVID-19 candidate vaccine Soberana 02 concluded March 31 in Havana with very good prospects as to safety and the immune response.
The statement was made on Cuban television by Doctor Maria Eugenia Toledo, leading scientific researcher of the clinical trial. Thus far results are very encouraging, said Toledo and noted that it has been a complex project which involved 44 thousand 10 Havana citizens who participated in the trial over the past 18 days.
The trial continues since it includes two and three doses of the vaccine over the next weeks, the scientists said and went on to explain that that the administration of any vaccine can always produces expected reactions, like mild pain and swelling on the injected body area, fever and some people feeling generally unwell.
Parallel to the phase III trials, the vaccine is being administered under an interventional study to the health personnel in Havana. In this regard, the doctor said that all the Havana population will be progressively receiving the shot since the capital city reports the largest COVID-19 prevalence.










Nos reservamos el derecho de no publicar los comentario que incumplan con las normas de este sitio